So far, we have discussed phase diagrams in relatively qualitative terms. The lever law is the primary way in which phase diagrams can be used to gain quantitative information about microstructures. With it, one can determine the fraction of each phase in a mixed microstructure and the composition of each phase present in the microstructure.
Consider the copper (Cu)-zinc (Zn) phase diagram below and a point on this diagram at a composition of 70 at% Zn and a temperature of 770 C, the red point on the phase diagram. The first step in using the lever law is to draw a horizontal line, called a "tie line" from this point until it intersects a single phase field on each end, as shown below. For the temperature and composition given on this diagram, the phase fields intersected by the tie line are Cu5Zn8 and liquid. The composition of each of these phases within the Cu5Zn8-liquid mixture is the overall composition at which the tie line intersects the phase field. These compositions are referred to as the "phase compositions." In the case stated above the phase composition of the Cu5Zn8 phase is 65 at% Cu and the composition of the liquid phase is about 74 at%.
Consider the copper (Cu)-zinc (Zn) phase diagram below and a point on this diagram at a composition of 70 at% Zn and a temperature of 770 C, the red point on the phase diagram. The first step in using the lever law is to draw a horizontal line, called a "tie line" from this point until it intersects a single phase field on each end, as shown below. For the temperature and composition given on this diagram, the phase fields intersected by the tie line are Cu5Zn8 and liquid. The composition of each of these phases within the Cu5Zn8-liquid mixture is the overall composition at which the tie line intersects the phase field. These compositions are referred to as the "phase compositions." In the case stated above the phase composition of the Cu5Zn8 phase is 65 at% Cu and the composition of the liquid phase is about 74 at%.
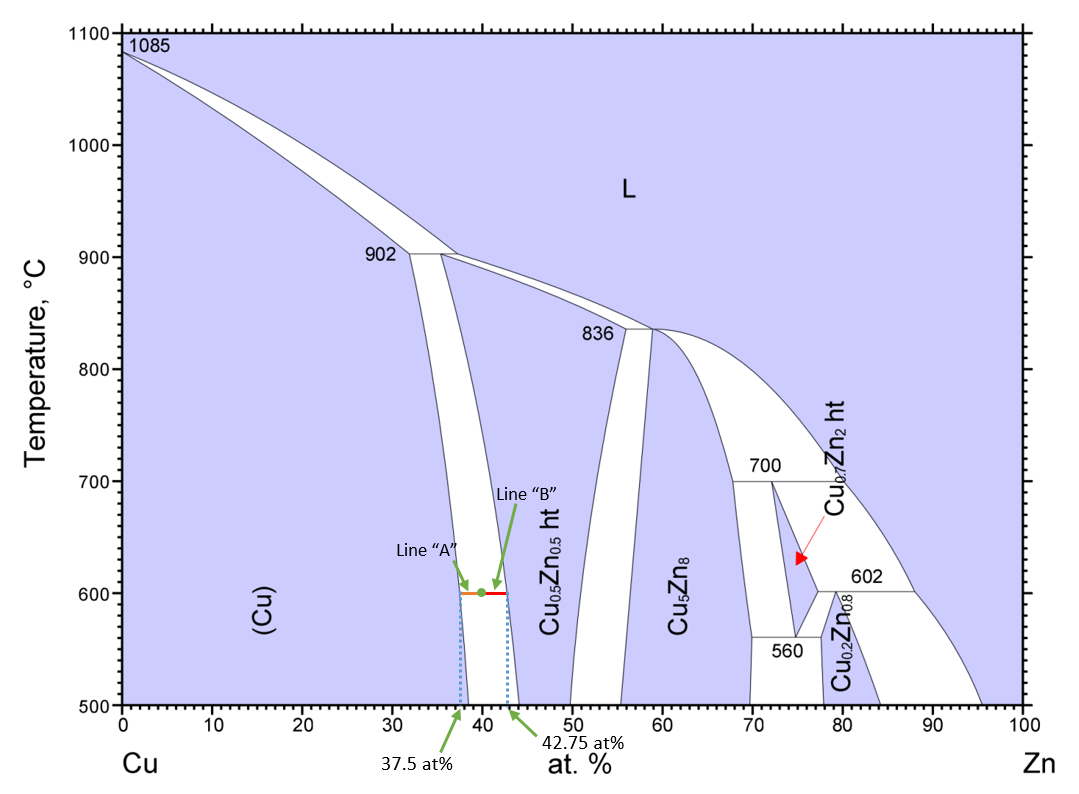
The other value that the lever law will help find is referred to as the "phase fraction." This is often confused with phase composition, so hopefully this will clear it up a bit: phase composition can best be thought of as how much of a species in in phase X. Phase fraction is how much of phase X is in a sample with overall composition X. To demonstrate how to calculate phase fractions, we'll consider the copper (Cu)-zinc (Zn) phase diagram again, but at an overall composition of 40 at% and temperature of 600C. The first step is the same as discussed above: the tie line should be drawn and the phase compositions noted. The "total length" of the tie line is the difference between these phase compositions. In order to determine the phase composition, the length of the line from the phase composition opposite the phase you are finding the fraction of to your overall composition is divided by the "total length" of the tie line.
The figure below shows the tie line and phase compositions at 40 at% Zn and 600C. The phase fraction of copper is then the length of line "B" over the total length of the tie line, (42.75-40)/(42.75-37.5)=0.52 or 52% copper phase. The phase fraction of the Cu0.5Zn0.5 phase is then the length of line "A" over the total length, or (40-37.5)/(42.75-37.5)=0.48 or 48%. This could also be calculated as 1-0.52=0.48 since the phase fractions must add up to 1, since there are only 2 phases present.
The figure below shows the tie line and phase compositions at 40 at% Zn and 600C. The phase fraction of copper is then the length of line "B" over the total length of the tie line, (42.75-40)/(42.75-37.5)=0.52 or 52% copper phase. The phase fraction of the Cu0.5Zn0.5 phase is then the length of line "A" over the total length, or (40-37.5)/(42.75-37.5)=0.48 or 48%. This could also be calculated as 1-0.52=0.48 since the phase fractions must add up to 1, since there are only 2 phases present.
There are two really common mistakes often made when using the lever law for this type of question. The first is confusing the definitions of phase composition and phase fraction. Remember: phase composition is the composition of each individual phase in percentage of the element or species being added. Phase fraction is the amount of each phase present in the microstructure. The other common mistake to avoid is using the wrong line lengths when calculating phase fractions. Often, the length of the line adjacent to the phase is mistakenly used instead of the length opposite. A little thought experiment to explain why the opposite length is used: suppose we have the Cu-Zn phase diagram above, again at 600C, but now at 38 at% Cu. The overall composition is very close to that of the copper phase, so it makes sense that the phase fraction of the copper should be nearly 100%. The length of the line from the phase composition of copper to the overall composition, however, is very short relative to the total length of the line, so using this would result in almost none of the copper phase in the microstructure, which doesn't make sense. That is why the length of the line opposite the phase is used.